Ions are electrically charged particles formed when atoms lose or gain electrons. They have the same electronic structures as noble gases.
Metal atoms form positive ions, while non-metal atoms form negative ions. The strong electrostatic forces of attraction between oppositely charged ions are called ionic bonds.
Ionic compounds have high melting and boiling points.
How ions form
Ions are electrically charged particles formed when atoms lose or gain electrons. This loss or gain leaves a complete highest energy level, so the electronic structure of an ion is the same as that of a noble gas - such as a helium, neon or argon.
Metal atoms and non-metal atoms go in opposite directions when they ionise:
Metal atoms form positive ions, while non-metal atoms form negative ions. The strong electrostatic forces of attraction between oppositely charged ions are called ionic bonds.
Ionic compounds have high melting and boiling points.
How ions form
Ions are electrically charged particles formed when atoms lose or gain electrons. This loss or gain leaves a complete highest energy level, so the electronic structure of an ion is the same as that of a noble gas - such as a helium, neon or argon.
Metal atoms and non-metal atoms go in opposite directions when they ionise:
- Metal atoms lose the electron, or electrons, in their highest energy level and become positively charged ions.
- Non-metal atoms gain an electron, or electrons, from another atom tobecome negatively charged ions.
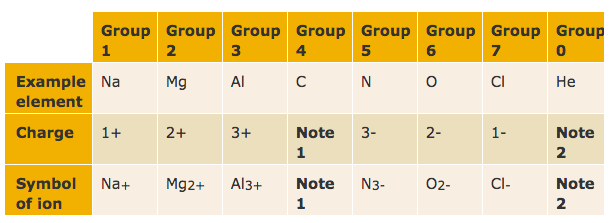
How many charges?
There is a quick way to work out what the charge on an ion should be:
There is a quick way to work out what the charge on an ion should be:
- the number of charges on an ion formed by a metal is equal to the group number of the metal
- the number of charges on an ion formed by a non-metal is equal to the group number minus eight
- hydrogen forms H+ ions
Metal ions
You need to be able to show the electronic structure of some common metal ions, using diagrams like these:
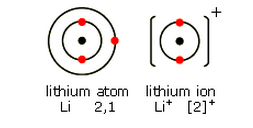
Lithium, Li
You need to be able to show the electronic structure of some common metal ions, using diagrams like these:
Lithium, Li
Lithium is in Group 1. It has one electron in its highest energy level. When this electron is lost, a lithium ion Li+ is formed.
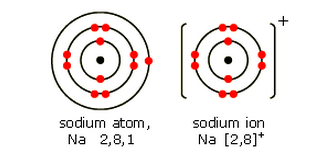
Sodium, Na
Sodium, Na
Sodium is also in Group 1. It has one electron in its highest energy level. When this electron is lost, a sodium ion Na+ is formed.
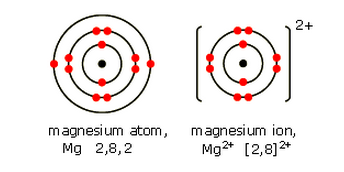
Magnesium is in Group 2. It has two electrons in its highest energy level. When these electrons are lost, a magnesium ion Mg2+ is formed.
A magnesium ion has the same electronic structure as a neon atom (Ne).
A magnesium ion has the same electronic structure as a neon atom (Ne).
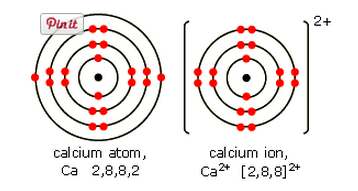
Calcium, Ca
Calcium is also in Group 2. It has two electrons in its highest energy level. When these electrons are lost, a calcium ion Ca2+ is formed.
A calcium ion has the same electronic structure as an argon atom (Ar).
A calcium ion has the same electronic structure as an argon atom (Ar).
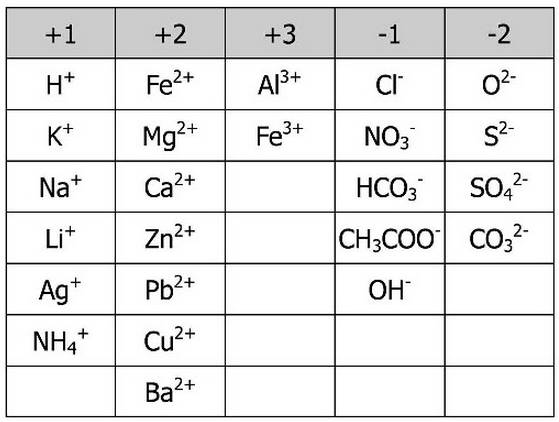
MONO ATOMIC AND POLYATOMIC IONS
There are two types of cation and anion. These are called MONO-ATOMIC and POLY-ATOMIC.
MONO mean 1. Mono-atomic ions are made of of only ONE type of atom.
POLY means many. Poly-atomic ions are charged particles, made of 2 or more atoms chemically bonded together.
MONO mean 1. Mono-atomic ions are made of of only ONE type of atom.
POLY means many. Poly-atomic ions are charged particles, made of 2 or more atoms chemically bonded together.